It is very likely that after a COVID-19 infection, even in cases that required no hospitalization, there may be some damage to the heart, specifically in the heart muscle, which may be damaged, fibrous, swollen, or scarred. This is the reality that studies show, and we cannot hide it.
We recommend a cardiovascular checkup before resuming sports and physical activities and no high-intensity workouts for those who’ve had the disease. We also recommend moderate-intensity maintenance workouts to avoid triggering potentially very serious and even life-threatening clinical pictures, in case the heart is compromised in ways we are not aware of.
Dr. Alfonso Galán González – Neolife Medical Team
When infected with COVID-19, the cells of the heart muscle suffer enormous damage. This has been confirmed with heart samples from patients who died of COVID-19.
As time goes by, we discover new aspects of the coronavirus disease. In this case, we will focus on the consequences, the residual damage it may have left in our body as it suffered the disease. We had anecdotal information from patients who had been sick at home almost without symptoms, but in which a chest CT scan showed pulmonary alterations that are typical in COVID-19, or cases of sudden deaths in patients who resumed physical activity after the disease had passed.
We wish to present the information and articles published in this regard and, above all, a very recent study that shows very clearly the kind of heart damage that this new virus may cause and its implications when it comes to resuming physical activity.
An article published in JAMA in September 2020 by Rajpal et al. presents the study of the hearts of 26 university athletes through cardiovascular nuclear magnetic resonance (CMR), individuals who’d had the COVID-19 disease without the need for hospitalization or any treatment beyond the management of the symptoms; 12 of them had had mild symptoms and the rest no symptoms at all. Four athletes (15%) presented CMR findings that were compatible with myocarditis, a disease in which the heart muscle is swollen and which may manifest as chest pain, heart failure, or sudden death; two patients had signs of pericarditis, fluid and inflammation in the membrane surrounding the heart; and 12 athletes (46%) had LGE (late gadolinium enhancement). This is a measure that is obtained in the CMR that shows us the overall condition of the heart muscle and whether there is fibrosis and scaring.
Another study, so recent that it is still pending review, conducted at the Gladstone Institute in San Francisco has shown experimentally the tremendous changes that occur when heart muscle cells are inoculated with COVID-19, and this has been confirmed with heart samples from patients who died of COVID.
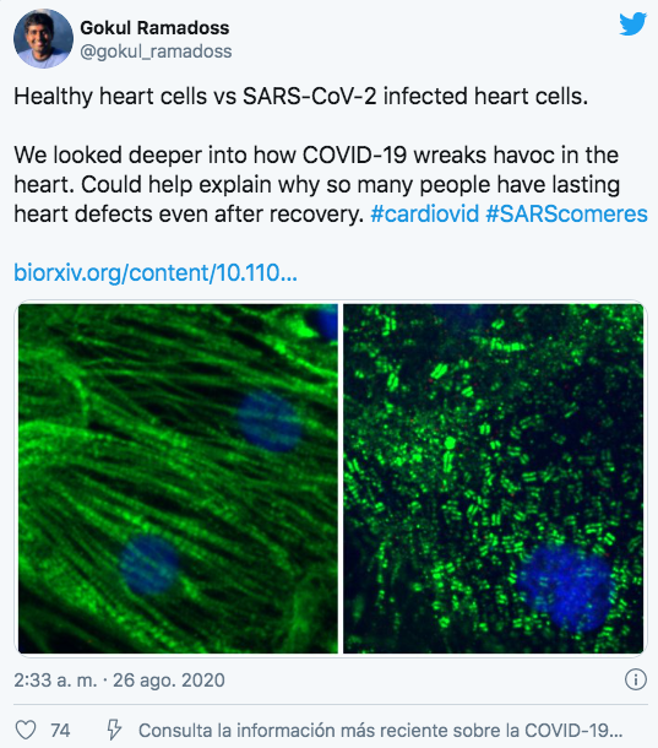
The fundamental study I wish to review in this blog post is going to require a little more explanation. It is a German study that looked into the level of heart damage caused by COVID-19 in randomly selected patients who tested positive for COVID-19 in PCR tests.
The study included 100 patients, 53 men and 47 women, with an average age of 49. They were given a CMR, and the level of troponin was measured (a marker of myocardial damage) an average of 71 days after the diagnosis of the disease.
Of the 100 patients, 67% recovered at home and 33% required hospital admission. A control group was set up matching the age group and risk factors, made up of individuals who had not had the disease.
We’ll present their findings, first as raw data, and then we’ll explain what these biomarkers mean:
- At the time of the assessment, high-sensitivity troponin T (hsTnT) levels were detected in 71 patients and were significantly high in 5.
- Compared to the healthy control group, patients who’d had COVID-19 had a lower left ventricular ejection fraction, higher left ventricular volumes, and changes in the CMR. Of these patients, 78% presented changes in the CMR, a 73% increase in native T1, a 60% increase in native T2, a 32% LGE, and a 22% increase in pericardial intensity. These results in terms of native T1 and troponin differed in a small but significant way among those who had recovered at home vs those who had needed hospitalization, with higher values for those hospitalized at the time, but not for native T2.
- The cardiac biopsy of those patients with more severe findings showed active white blood cell inflammation.
Well, all this sounds very interesting, but what does it all mean? What do these measures mean? Are they just strange laboratory values that have nothing to do with the patient’s prognosis or clinical symptoms?
The LGE is, as we have already mentioned, a marker of myocardial damage of a different nature, and if associated with pericardial effusion, it may be attributed to fibrosis and/or edema due to ongoing active pericarditis. If we have fluid in the membrane that covers our heart, its ability to relax is compromised.
Increased measures of native T1 represent diffuse myocardial fibrosis and/or edema, while native T2 is specific to edema. Therefore, patients with higher native T1 and T2 levels are undergoing an active inflammatory process, while those with higher native T1 levels and normal native T2 levels have healed with some residual diffuse myocardial damage.
Higher levels of native T1 have been associated with a worse prognosis in patients with ischemic heart disease (coronary obstruction) and non-ischemic cardiomyopathies.
Higher levels of troponin and C-reactive protein indicate ongoing myocardial damage and inflammation respectively and have been associated with a worse prognosis even if the increase is low.
However, we may conclude that this study has revealed heart damage in 78 patients (78%) and ongoing myocardial inflammation in 60 patients (60%), regardless of pre-existing conditions, the severity and overall course of acute disease and time since the original diagnosis.
Other studies have placed this at 50% and others at less. But even if this figure was actually a little lower than this 78%, we are talking about huge percentages. It is very likely that after a COVID-19 infection, even in cases that required no hospitalization, there may be some damage to the heart, specifically in the heart muscle, which may be damaged, fibrous, swollen, or scarred. This is the reality that studies show, and we cannot hide it.
Before we knew about these findings, all of us doctors heard from our patients how hard it was for them to resume their exercise routines after having COVID-19, how tired they were, the discomfort they felt while doing an activity that they used to do without any problems and that they used to enjoy. In view of this data, we can no longer simply say this virus makes us tired, like other viruses do, like the CMV, flu, or Epstein Barr do.
We strongly recommend getting a cardiovascular checkup before resuming sports and physical activity. The British Journal of Sports Medicine gave the following recommendations in April. With what we know and has been published since then, perhaps the CMR may have to be included sooner rather than later.

We also recommend avoiding high-intensity workouts if you’ve had the disease. The current recommendation is moderate-intensity maintenance workouts to avoid triggering potentially very serious and even life-threatening clinical pictures, in case the heart is compromised in ways we are not aware of.
BIBLIOGRAPHY
(1) Juan A. Pérez-Bermejo, Serah Kang, Sarah J. Rockwood, Camille R. Simoneau, David A. Joy, Gokul N. Ramadoss, Ana C. Silva, Will. Flanigan, Huihui Li, Ken Nakamura, Jeffrey D. Whitman, Melanie Ott, Bruce R. Conklin, Todd C. McDevittSARS-CoV-2 infection of human iPSC-derived cardiac cells predicts novel cytopathic features in hearts of COVID-19 patients. bioRxiv 2020.08.25.265561
(2) Rajpal S, Tong MS, Borchers J, et al. Cardiovascular Magnetic Resonance Findings in Competitive Athletes Recovering From COVID-19 Infection. JAMA Cardiol. Published online September 11, 2020.
(3) https://blogs.bmj.com/bjsm/2020/04/24/the-resurgence-of-sport-in-the-wake-of-covid-19-cardiac-considerations-in-competitive-athletes/
(4) Puntmann VO, Carerj ML, Wieters I, et al. Outcomes of Cardiovascular Magnetic Resonance Imaging in Patients Recently Recovered From Coronavirus Disease 2019 (COVID-19) [published online ahead of print, 2020 Jul 27]. JAMA Cardiol. 2020;e203557. doi:10.1001/jamacardio.2020.3557
(5) https://www.femede.es/documentos/Reconocimientos_medicos-COVID19-01.pdf
