The mTOR and AMPk metabolic pathways are of paramount importance for the control of metabolism and its two classic phases, anabolism (tissue creation) and catabolism (its destruction) but also in the development of pathologies. By knowing what they are and studying them, we may obtain therapeutic weapons against cancer or diabetes.
The AMPk pathway acts more like an energy sensor in our body. It detects when we have ATP deficiency (our energy currency) and sets into motion the mechanisms to get it back through the oxidation of fatty acids in muscle and liver, improving insulin resistance and glucose entry into the muscle, and inhibiting triglyceride synthesis.
Dr. Alfonso Galán González – Neolife Medical Team
Over-activation of the mTOR pathway may lead to cancer, insulin resistance, diabetes, Alzheimer’s and Parkinson’s, among other diseases.
As promised in our previous blog posts, we will go into greater detail about these two metabolic pathways, the AMPk and mTOR, both of paramount importance in the control of metabolism and its two phases, anabolism (tissue creation) and catabolism (its destruction). But they also have a huge influence on pathology, and we’re not talking about strange pathologies we have never heard of before, but rather diseases like cancer, diabetes, cardiovascular disease, Alzheimer’s, Parkinson’s, etc.
Is your interest piqued? Read on and find out how you can achieve a balance between these pathways to live a longer, healthier life (Xu J et al. 2012).
mTOR
This metabolic pathway is formed by a series of proteins that regulate processes as important as tissue creation through protein synthesis and cellular hyperplasia. It is therefore a fundamentally anabolic pathway. Its name comes from the discovery made regarding its relationship with Rapamycin. The inhibition of this pathway with Rapamycin leads to increased longevity (Mammalian Target Of Rapamycin). In addition to this drug, it is regulated by other factors such as insulin, IGF-1 (insulin-like Growth Factor-1), amino acids (leucine), strength training, etc.
Indeed, the inhibition of the mTOR pathway has been linked to an increase in the longevity of certain organisms (Johnson SC et al. 2013). Apparently, on a biological level, tissue creation is at odds with longevity. Thus, fast-growing animals have a short life expectancy (see, for example, a mouse vs an elephant). Increased protein synthesis, the creation of tissue. and cell hyperplasia are beneficial during youth to help the body grow, but subsequently keeping this pathway activated would have negative effects on longevity. Does this mean that we must always inhibit it? No. Balance is the important thing, as we said at the start. There are specific situations where it’s necessary to activate it, and we will go over them below.
A carbohydrate-rich diet with the consequent elevation of insulin levels, the availability of amino acids and IGF-1 activate this pathway, while calorie restriction (which we discussed extensively in other articles), a lower intake of amino acids, especially leucine and the aforementioned Rapamycin would inactivate it.
Over-activation of the mTOR pathway may lead to cancer, insulin resistance, diabetes, Alzheimer’s, and Parkinson’s, among other diseases (Saxton RA et al. 2017).
In relation to cancer, the mTOR pathway is complex:
- Activating this pathway through the above mechanisms causes oncogenes to be activated.
- By inhibiting autophagy, our system’s ability to attack tumor cells is also altered.
- The processes of angiogenesis are activated, that is, the creation of new blood vessels that provide blood with nutrients to the tumor.
- Changes in the energy metabolism of cells cause them to burn energy in the absence of oxygen (anaerobic glycolysis), typical of tumor cells.
As for its relationship with Alzheimer’s and Parkinson’s, the formation of protein agglomeration typical of these diseases (neurofibrillary tangles and B-amyloid plaques) is favored by the activation of mTOR, as it leads to the formation of proteins and inhibits autophagy (which is the mechanism by which the cell would “recycle” protein agglomeration).
So what can be good about mTOR based on the above?
Activating this pathway in certain situations or moments is very beneficial, as well. We need to have tissue creation and muscle synthesis to repair and develop muscle after exercise, so as not to lose muscle mass over the years (sarcopenia), to heal after injuries or surgical interventions, etc.
AMPk
This pathway acts more like an energy sensor in our body. It detects when we have ATP deficiency (our energy currency) and sets into motion the mechanisms to get it back through the oxidation of fatty acids in muscle and liver, improving insulin resistance and glucose entry into the muscle, inhibiting triglyceride synthesis, cholesterol, and the accumulation of fat in adipocytes, etc. (Lin SC et al. 2018).
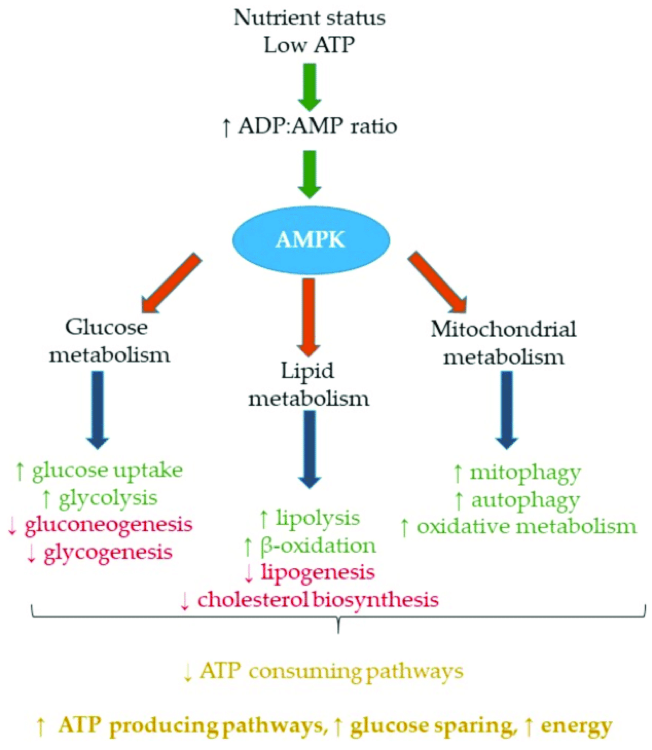
What activates the AMPk pathway?
-
Anything that lowers ATP concentrations and increases those of AMP (product of the use of ATP for energy) and situations that lead the cell to a situation of stress (oxidative stress with increased reactive oxygen species and free radicals, hypoxia, lack of glucose, etc.) may lead to an increase in the AMPk pathway.
-
Exercise, especially high intensity (HIIT workouts, Tabata) causes this pathway to be activated to obtain energy for its performance. This explains part of the effects of exercise on fat burning and its effect on the increase of insulin sensitivity (Thompson DM et al. 2018).
-
Several substances have been investigated for this purpose. We will mention, as the most widely recognized and used in practice, metformin, berberine, resveratrol, curcumin, alpha-lipoic acid, and caffeine.
- Fasting, as a form of calorie restriction, is mainly implemented as intermittent fasting. After 10 to 12 hours of fasting, this pathway is already activated.
The effects of the activation of this pathway are numerous, very important, and varied, as we have already mentioned.
- By improving insulin resistance, it prevents the development of type 2 diabetes.
- It prevents and improves fatty liver (nonalcoholic hepatic steatosis), associated with increased visceral fat, by promoting the oxidation of fatty acids and inhibiting fat deposit in adipocytes.
- It promotes mitochondrial biosynthesis and oxidative metabolism at this level. In other words, it helps the power plant in our cells, the mitochondria, work better and even lets us have more of them to be able to obtain more energy and more efficiently. This, at the heart muscle level, inhibits the processes of cell death in response to cardiac ischemia and prevents heart damage in these situations.
- It is related to other hormones that are important for energy production, such as catecholamines (adrenaline and norepinephrine), cortisol, thyroid hormones T4 and T3 (also encouraging the conversion of T4 into T3, which is the active hormone), glucagon, growth hormone, and increases the production of sex hormones (Bertoldo MJ et al. 2015).
- It lowers inflammation by directly inhibiting NFKB, an inflammation activator. But chronic inflammatory states tend to inhibit it (Jeon SM et al. 2016).
- In polycystic ovary syndrome, which is much more frequent than we think and in which the woman can suffer a series of alterations such as infertility, hirsutism, obesity, ovarian cysts, etc., above all else, what is most constant in this syndrome is insulin resistance, which, as we have seen, can improve by activating the AMPk pathway. Therefore, these women improve greatly with metformin.
- The relationship between AMPk and cancer has been researched for over 17 years when its connection with tumor suppression genes (anti-oncogenes), such as LKB1 and p53 (Ciccarese F et al. 2019) was discovered. It was proven that activating this pathway may prevent some cancers or slow their growth. But, like everything, the relationship is more complex, involving autophagy and the previously mentioned change in energy metabolism shown by tumor cells, as well as phenomena of resistance to chemotherapy, where it seems that the role of AMPk may be the opposite, favoring said resistance by helping tumor cells resist the stress they are suffering (Wang Z et al. 2016).
Therefore, much remains to be investigated and discovered in this very important area. It does seem very promising in the treatment of this enormous scourge that is cancer.
And now we close this first part of the explanation of these interesting metabolic pathways. In the second part, we will propose ways to balance them to have the best of both and avoid the negative effects of either the activation or inhibition of each.
BIBLIOGRAPHY
(1) Thomson DM. The Role of AMPK in the Regulation of Skeletal Muscle Size, Hypertrophy, and Regeneration. Int J Mol Sci. 2018;19(10):3125. Published 2018 Oct 11. doi:10.3390/ijms19103125
(2) Lin SC, Hardie DG. AMPK: Sensing Glucose as well as Cellular Energy Status. Cell Metab. 2018;27(2):299‐313. doi:10.1016/j.cmet.2017.10.009
(3) Ciccarese F, Zulato E, Indraccolo S. LKB1/AMPK Pathway and Drug Response in Cancer: A Therapeutic Perspective. Oxid Med Cell Longev. 2019;2019:8730816. Published 2019 Oct 31. doi:10.1155/2019/8730816
(4) Wang Z, Wang N, Liu P, Xie X. AMPK and Cancer. Exp Suppl. 2016;107:203‐226. doi:10.1007/978-3-319-43589-3_9
(5) Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease [published correction appears in Cell. 2017 Apr 6;169(2):361-371]. Cell. 2017;168(6):960‐976. doi:10.1016/j.cell.2017.02.004
(6) Bertoldo MJ, Faure M, Dupont J, Froment P. AMPK: a master energy regulator for gonadal function. Front Neurosci. 2015;9:235. Published 2015 Jul 14. doi:10.3389/fnins.2015.00235
(7) Jeon SM. Regulation and function of AMPK in physiology and diseases. Exp Mol Med. 2016;48(7):e245. Published 2016 Jul 15. doi:10.1038/emm.2016.81
(8) Xu J, Ji J, Yan XH. Cross-talk between AMPK and mTOR in regulating energy balance. Crit Rev Food Sci Nutr. 2012;52(5):373‐381. doi:10.1080/10408398.2010.500245
(9) Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013;493(7432):338‐345. doi:10.1038/nature11861
(10) Kim J, Yang G, Kim Y, Kim J, Ha J. AMPK activators: mechanisms of action and physiological activities. Exp Mol Med. 2016;48(4):e224. Published 2016 Apr 1. doi:10.1038/emm.2016.16
(11) Basualto-Alarcón C, Jorquera G, Altamirano F, Jaimovich E, Estrada M. Testosterone signals through mTOR and androgen receptor to induce muscle hypertrophy. Med Sci Sports Exerc. 2013;45(9):1712‐1720. doi:10.1249/MSS.0b013e31828cf5f3

